 |
 |
The Influenza Virus Hemagglutinin
The world has been aware of the disease and symptoms of influenza for centuries with periodic epidemics recorded in the 19th and 20th centuries. The flu pandemic of 1918-1919 led to an estimated 20 million deaths around the world - more than the number killed in the battles of World War I. The virus, itself, was first isolated in the 1930's and the first glimpse of its structure came in 1943 when it was seen in the electron microscope.
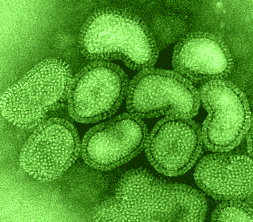 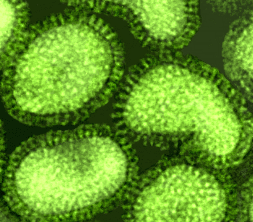 images courtesy of Linda M. Stannard, University of Cape Town, |
 The outer surface of the virus contains two glycoproteins, the hemagglutinin
and the neuraminidase embedded in the virus lipid membrane envelope. The hemagglutinin
or HA was so named because it is the protein responsible for the ability of
flu virus to agglutinate red blood cells and for the binding of the virus to
cells via its attachment to sialic acid. It is now recognized as the major virulence
factor associated with this virus.
The outer surface of the virus contains two glycoproteins, the hemagglutinin
and the neuraminidase embedded in the virus lipid membrane envelope. The hemagglutinin
or HA was so named because it is the protein responsible for the ability of
flu virus to agglutinate red blood cells and for the binding of the virus to
cells via its attachment to sialic acid. It is now recognized as the major virulence
factor associated with this virus.
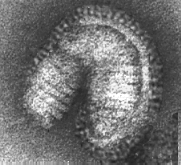
In 1981 Wilson, Wiley and Skehel published the atomic structure of the hemagglutinin to a resolution of 3 angstroms – this was the first look at the structure of a viral membrane protein. Two years later, the structure of influenza virus neuraminidase to a resolution of 2.9 angstroms was solved by Varghese, Laver and Coleman.
John Skehel began his studies with the hemagglutinin in the early 1970s John Skehel Oral History and not long afterward, Don Wiley decided that this protein was the one whose structure he would determine. One of the deciding factors was the report by Brand and Skehel in 1972 that they had obtained crystals of a soluble form of the HA clipped from its membrane anchor by the proteinase, bromelain. Wiley Oral History Wiley obtained some of this purified HA from Skehel and began, but both Wiley and Skehel agree that the first crystals obtained at Harvard may really have been - pieces of broken glass! Wiley Oral History John Skehel Oral History. But that was only for a brief moment and Don was soon able to obtain beautiful crystals of the hemagglutinin that diffracted out to 3 Ĺ.
Don Wiley took a sabbatical in London where he worked with Skehel and this began their long term collaboration. He then returned to Harvard - Ian Wilson joined the lab and together they solved the structure of HA. Wiley Oral History It is an unusual structure: the protein is a trimer with two distinct regions - a stem consisting of a triple stranded coiled-coil of alpha-helices extending 76 Ĺ from the membrane and a globular region of antiparallel beta-sheet on top.
What did this tell us about the biological activities?
One satisfying result was to see that the host-receptor binding site was located on the top globular portion. It is the amino acids that surround this region that have undergone change in most of the antigenic variants appearing in influenza virus epidemics.
Even more interesting were the problems raised about the presumed role of HA in fusion. By the time the atomic structure was solved, HA was known to be the mediator of fusion between the virus and cell membranes. The amino terminus of HA2 was thought to be the fusion peptide but the atomic structure showed that it was tucked inside the stalk of the HA and was located 100 Ĺ from the tip and 35 Ĺ from the membrane end of the molecule. If this were the fusion peptide, how did it work? How could it insert into a cellular membrane? The length of the HA molecule was 135 Ĺ from the virus membrane to the receptor pocket on the globular head and this structure should restrict membranes from getting close enough together to fuse.
Don Wiley recalls that the critical experiment, the experiment that demonstrated that the hemagglutinin underwent a conformational change at low pH, was by John Skehel in 1982. Wiley Oral History
"In 1982, John made what I think is the discovery. He doesn't agree he thinks that it was the structure. He's the person who lowered the pH of some hemagglutinin and showed that the hemagglutinin underwent a conformational change that was clearly the equivalent of fusion in the sense that you took BHA (the bromelain treated HA) which was not a membrane protein, lowered the pH to the pH of the endosomes and it became a membrane protein. If you did it in the presence of lipid vesicles it stuck to the lipid vesicles without a transmembrane tail, or if you did it in the presence of detergent micelles it stuck to detergent micelles, or if you did it in the presence of nothing - water- it formed rosettes which you could see in the electron microscope and looked just like the hemagglutinin would look with its transmembrane tail before you lowered the pH. It looked like a membrane protein in water; namely, a protein-protein micelle so that really established I think the important thing."
And then Don said: "The paper was clear about it. Whether anyone else got it is another issue. We got it - believe me. I remember that one of the things we were worried about is - well, maybe the protein is just completely falling apart and we would talk everyday over the phone and I think it was me who had the 'brilliant' idea of asking whether it hemagglutinated after you lowered the pH. Because before you lowered the pH, BHA won't hemagglutinate because it's not rosettes but after you lowered the pH if it really stuck together as rosettes and it were really still the hemagglutinin and hadn't been shot in some way, then the binding sites should still be O.K. and it should hemagglutinate. Sure enough it did. So then we sort of knew that the molecule was undergoing a conformational change."
But in the 1982 paper the authors wrote: "The limited changes observed even after tryptic digestion argue against gross secondary structure modifications". They thought that the changes were "not inconsistent with relative movements of molecular domains that independently maintain their tertiary structure".
In the mid-80s Skehel and his colleagues measured substantial physical changes in the hemagglutinin after it had been exposed to low pH. Electron microscopy indicated that the molecule became thinner and elongated (Ruigrok et al. EMBO J 5: 41-49,1986). But at that time, the idea that proteins could undergo drastic conformational changes was not so easy to accept.
Don admits that his response to the electron microscopy was:
"No that's completely impossible, protein's don't unfold, this is a solid protein. Look at all this beta sheet, look at this helix holding on here and so on. This will never happen".
A structure for portions of the low pH form of HA2 was solved in 1994 and we now know that after exposure to low pH the conformational changes are very impressive: portions of the HA2 shift from an extended chain into an alpha helix and other sequences move from an alpha helix to an extended chain. In this new structure, the fusion peptide is at the tip of an elongated coiled-coil chain and the domains near the C-terminal interact with those near the amino terminus. The low pH form is more thermodynamically stable than the native form of the HA2. The initial folding of the HA2 domains leads to a metastable configuration. Exposure to acid converts this form to the thermodynamically stable form of the molecule. An implication of these alterations is that a protein may not always fold into its most stable conformation. Does this alter how we think about protein folding?
| Introduction | Some historical highlights: structural virology and virology |
| Solving the Structure of Icosahedral Plant Viruses | Picornavirus Structure | Poliovirus | Polio
The Influenza Virus Hemagglutinin | The Influenza Virus Neuraminidase |
Issues of Science and Society |contributors|
Home | Discussion Database |